|
12/30/2023 0 Comments Periodic table series Today’s primary use of the Periodic Table is education. A widely used version of the Periodic Table in 18-column format has been used since the 1950s and can trace its usage back to Deming’s General Chemistry (1923) and even before that to Alfred Wegner’s 1905 “block” layout. In summary, there is no official version of the Periodic Table approved by the International Union of Pure and Applied Chemistry (IUPAC) or other bodies. Even in our digital world, aspect ratios on web pages and computer monitors or smartphones typically favor the 18-column design. The 32-column format requires foldouts or much longer printed charts. The 18-column version is more compact with a favorable aspect ratio allowing it to fit easily on textbook pages or handouts. Indeed the 32-column format has multiple advantages however, it has a significant disadvantage: layout space. The 18-column version of the Periodic Table is not superior to other versions. The wide distribution of these materials in Western countries and the practical format led to this 18-column form becoming the most popular version. These continued to be provided with new editions for several decades. letter-size printouts as part of a promotional campaign. The long version with the f-block “inline” has 32 columns.ĭeming’s table achieved a breakthrough in 1928 because the publisher distributed U.S. It contains 18 columns and is itself derived from Alfred Wagner’s 1905 18-column “block” layout reflecting the s-, d-, and p-blocks (sub-shells). Deming’s 1923 General Chemistry textbook. Today’s most used version of the Periodic Table evolved from Horace G. Others were arrangements referred to as “rocket ships,” based on Niels Bohr’s early Periodic Tables. Some were 8-column, 18-column, and 32-column tables. There were several forms of the Periodic Table used in textbooks in the 1950s. The discovery of many more elements than originally in Mendeleev’s Table, increased understanding of the nucleus (protons and neutrons), as well as electron orbitals, led to the modern Periodic Table. Doctoral Dissertation, Creating a Symbol of Science: The Development of a Standard Periodic Table of the Elements Ann Robinson, p. “I believe that the chief difference is that you are using the periodic table to express the probable configuration of the electron shells, while I and a few other chemists are primarily concerned with the representation of the chemical character of the elements.” – F.A.Paneth to G.T.Seaborg, 14 July 1950, Box 342, Glenn Theodore Seaborg Papers, Manuscript Division, Library of Congress, Washington, D.C. 
Finally, Glenn Seaborg proposed the f-series (Actinide Series) based on his research on Americium and Curium. Combined with Pauli’s exclusion principle a quantum rule for filling electron shells was determined. This was further adapted into the Bohr model. This led to the octet rule and valence bond theories. In 1914 Johannes Rydberg determined a relationship in the atomic numbers of noble gases. Skipping ahead to 1913, Anton van den Broek, proposed the nuclear charge determined the placement in the Periodic Table. This 8-column format was used for decades, even after other formats were developed. By 1871 Mendeleev had a Periodic Table in eight groups related to oxidation states. Mendeleev’s gap for Gallium ( Eka-Aluminium) is a well-known example. Mendeleev left gaps where there was no known element matching the anticipated weight and properties. Frequently incorrect weights were reported and later revised when purer samples could be analyzed. He was correct because many elements were difficult to isolate in the samples at the time. Mendeleev trusted experimental evidence of chemical reactions more than the measured weights. 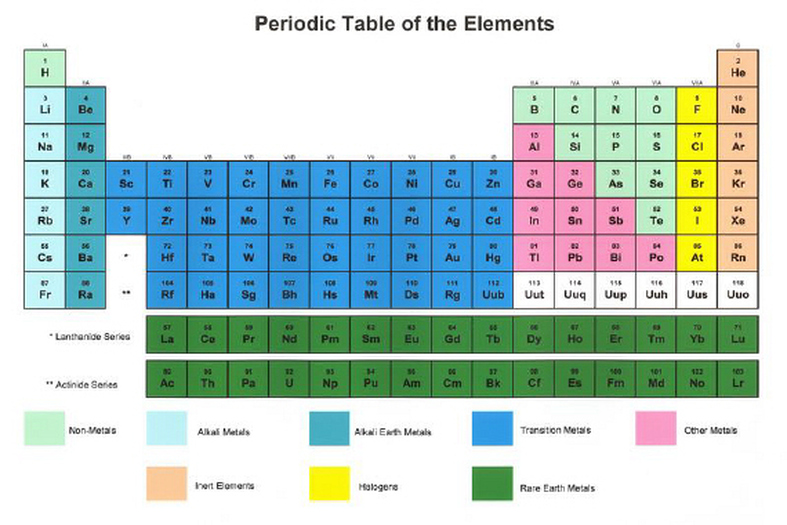
He used atomic weights to sort the elements however, he reordered them based on observed properties if they did not seem to be in the right spots. In 1863 Dmitri Mendeleev drafted the first of his 60 versions of the Periodic Table. 
He observed there were similar properties every eight elements, so he organized his table with eight columns. 
John Newlands organized elements based on weights. Let us take a brief look at both the history and the most common versions in use today. While the information about each element in the Periodic Table is the same, different formats have been used to organize the Periodic Table. Hmmm, is this element a Ravenclaw or Hufflepuff? Versions of the Periodic Table For Harry Potter fans, think of it as an algorithmic version of a sorting hat. The organization of the Periodic Table in Rows and Columns is not haphazard. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
